In 2013, Lopez-Otin and colleagues published what has become the most cited paper in ageing biology: "The Hallmarks of Aging," in Cell, which identified nine core cellular and molecular mechanisms that accumulate across the lifespan to collectively produce the physiological changes associated with ageing. The framework — updated to twelve hallmarks in a 2023 revision — transformed longevity research from a descriptive epidemiological enterprise into a mechanistic biology programme with specific molecular targets and, for the first time, a conceptual framework for evaluating whether a given nutritional or pharmacological intervention addresses the biological machinery of ageing rather than merely correlating with longer lifespan in observational data. The longevity supplement industry, which existed long before this framework, has largely continued to operate without it — citing individual compound studies without integrating them into a coherent understanding of which hallmarks are being addressed, which are not, and which interactions between compounds produce effects greater than the sum of their parts. This article applies the hallmarks framework systematically to the most evidence-supported longevity supplement compounds, maps each to the specific hallmarks it addresses at the molecular level, and explains how the Zenutri Longevity Plus Bundle was architected around this framework — providing the most comprehensive hallmark coverage available in a TGA-listed Australian-made daily protocol.
Key Takeaways
- Understand the Lopez-Otin hallmarks of ageing framework — the twelve cellular and molecular mechanisms that accumulate to produce the physiological changes of ageing — and identify which of the four most nutritionally addressable hallmarks (mitochondrial dysfunction, cellular senescence and SASP, epigenetic alterations, and deregulated nutrient sensing) are most directly relevant to the supplement decisions of Australian adults in the 40 to 65 age bracket.
- Learn the cellular senescence and SASP mechanism in detail — why senescent cells that accumulate with age secrete a pro-inflammatory mixture of TNF-α, IL-6, IL-1β, and MMP-1/3/9 (the senescence-associated secretory phenotype) that progressively impairs tissue function in every organ system, and how curcumin's IKK-β inhibition of NF-κB (SASP transcription) and resveratrol's SIRT1-mediated autophagy activation (senescent cell clearance) address the SASP burden through two independent and complementary mechanisms.
- Discover the mTOR/AMPK/SIRT1 nutrient sensing nexus — how chronic mTOR overactivation suppresses autophagy and accelerates the loss of proteostasis hallmark, how AMPK counteracts this through mTOR inhibition, and how resveratrol's SIRT1 activation produces the indirect mTOR/AMPK-mediated autophagy induction that makes the resveratrol-NR co-formulation in Reversa NR (AUST L 520794) more than the sum of its individual compound effects.
- Understand the epigenetic drift hallmark — the progressive loss of the epigenetic marks (DNA methylation patterns, histone acetylation states) that determine gene expression programmes in differentiated cells — and how SIRT1's deacetylase activity on histones H3K9, H3K14, and H4K16 provides the epigenetic maintenance function that partially counteracts this hallmark, making SIRT1 activation via resveratrol in CurcuNova (AUST L 520796) and Reversa NR a longevity mechanism beyond the energy and anti-inflammatory effects these compounds are primarily discussed for.
- Navigate the complete Zenutri Longevity Plus Bundle architecture — Reversa NR (AUST L 520794) for NAD+-SIRT1-mitochondrial dysfunction and nutrient sensing; UbiQ Forte (AUST L 520795) for ETC electron carrier function and CoQ10-specific mitochondrial membrane protection; CurcuNova (AUST L 520796) for NF-κB-SASP inhibition, Nrf2-antioxidant upregulation, and SIRT1 epigenetic maintenance; and Osteo+Core (AUST L 520792) for D3/K2 calcium metabolism and the vascular calcification prevention that protects the cardiovascular system from the matrix Gla protein-related calcification that K2 insufficiency accelerates — understanding how each formulation addresses a distinct and non-overlapping set of hallmarks.
The Hallmarks of Ageing: The Molecular Targets Longevity Science Is Converging On
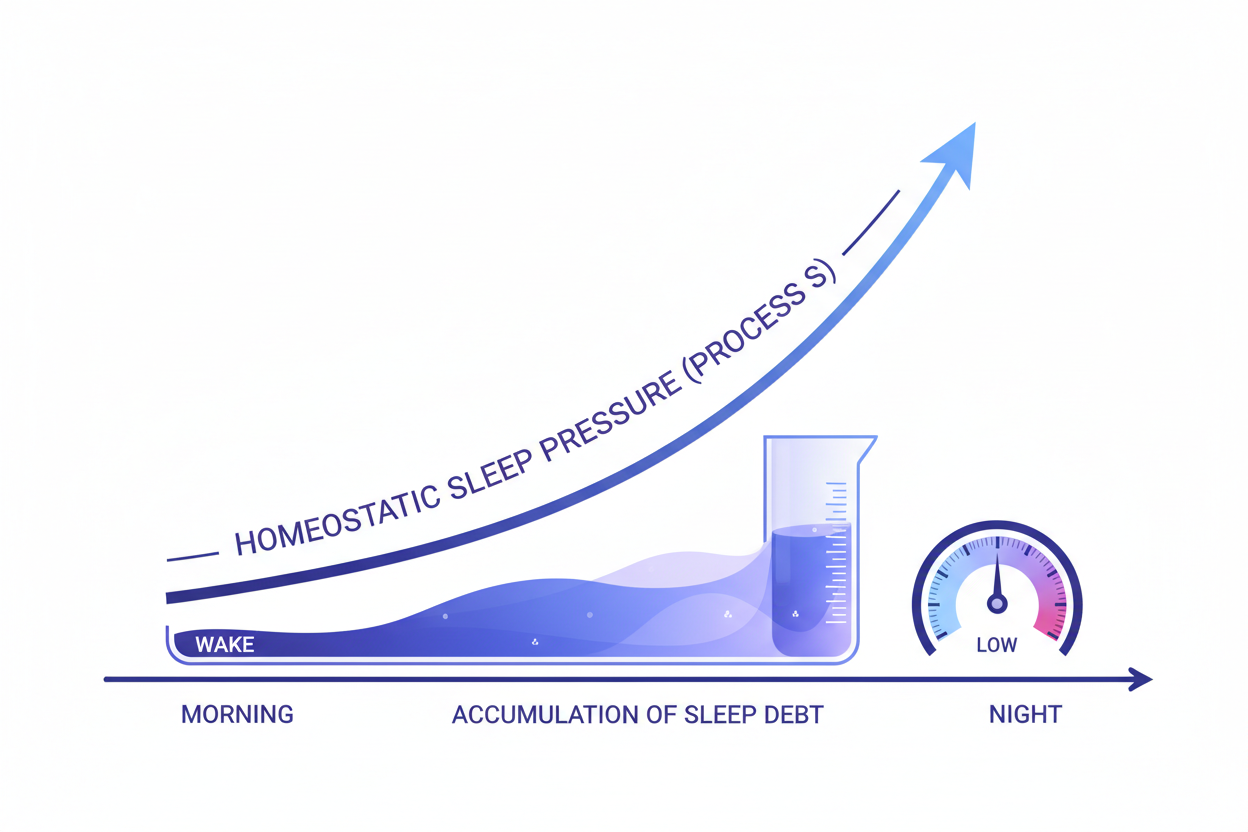
The twelve hallmarks of ageing identified in the Lopez-Otin 2023 framework revision represent the current consensus of what the molecular biology of ageing looks like at the cellular level. They are divided into three tiers. The "primary" hallmarks are the initial damage-generating events: genomic instability, telomere attrition, epigenetic alterations, and loss of proteostasis. The "antagonistic" hallmarks are responses to primary damage that are initially protective but become harmful with overactivation: deregulated nutrient sensing, mitochondrial dysfunction, and cellular senescence. The "integrative" hallmarks are the downstream tissue and systemic consequences of accumulated primary and antagonistic hallmark effects: stem cell exhaustion, altered intercellular communication, disabled macroautophagy, chronic inflammation, and dysbiosis. The framework's clinical utility for supplement selection is that it identifies which cellular mechanisms a given compound addresses, whether those mechanisms correspond to primary or downstream hallmarks, and how combinations of compounds can address multiple hallmarks simultaneously with non-overlapping mechanisms.
The four hallmarks most directly addressable through the nutritional interventions available in the Australian supplement market are: mitochondrial dysfunction (the progressive decline in electron transport chain efficiency, NAD+ availability, and mitochondrial biogenesis that is the most direct nutritional target); cellular senescence and the SASP (the accumulation of senescent cells and their pro-inflammatory secretory programme, directly targeted by NF-κB inhibitors and SIRT1 activators); epigenetic alterations (the progressive drift from the epigenetic programmes established in development, partially counteracted by SIRT1 histone deacetylase activity); and deregulated nutrient sensing (the chronic mTOR overactivation and AMPK underactivation of the Western dietary pattern, addressable through SIRT1-mediated nutrient sensing modulation). The D3/K2 combination addresses a fifth domain not in the original hallmarks framework but directly relevant to the cardiovascular and skeletal consequences of ageing: the calcium metabolism dysfunction that produces both bone mineral density loss and vascular calcification as the same nutrient imbalance produces excess soft-tissue calcium deposition while failing to direct it to bone matrix. To understand which combination of these pathways is most relevant to your specific biological context, take the Zenutri personalised health quiz.
Healthspan Versus Lifespan: The Goal That Modern Longevity Supplements Actually Target
The distinction between lifespan (chronological years of life) and healthspan (years of life spent in good health, with maintained physical and cognitive function, metabolic resilience, and freedom from significant chronic disease burden) is the most important clarification in longevity supplement research. The compounds most studied in the longevity field — resveratrol, NAD+ precursors, CoQ10, polyphenols, D3/K2 — do not extend maximum lifespan in the dramatic sense that the "anti-ageing supplement" category implies. What the best-powered human clinical evidence demonstrates for these compounds is improvement in the specific physiological parameters that determine healthspan: mitochondrial energy efficiency, cardiovascular function, bone mineral density maintenance, inflammatory burden, and the cellular quality control mechanisms that prevent the progressive accumulation of damaged components that drives functional decline. Pursuing healthspan extension through nutritional intervention is both scientifically credible and appropriately modest — the goal is not to live to 120, but to maintain the physical and cognitive capacity that allows a full, active life through the 60s, 70s, and 80s that the longevity revolution is increasingly making available to the Australian population.
Mitochondrial Dysfunction: The Energy Decline Hallmark
Mitochondrial dysfunction is simultaneously the hallmark most directly addressable through nutritional supplementation and the hallmark whose effects are most immediately felt at the functional level, because the mitochondrial energy decline of ageing is the cellular substrate of the fatigue, reduced exercise capacity, and cognitive performance changes that Australian adults notice most clearly as they move through their 40s and 50s. As covered in the supplements for energy and fatigue article in this series, the primary mechanisms of age-related mitochondrial dysfunction are NAD+ decline (reducing Complex I electron transport chain throughput), CoQ10 depletion (reducing the electron carrier availability between Complex I/II and Complex III), progressive oxidative damage to mitochondrial membrane lipids and enzyme complexes, and the decline in mitochondrial biogenesis driven by reduced PGC-1α activity. The hallmarks framework adds a critical dimension: mitochondrial dysfunction is not merely an energy-production problem but also a driver of multiple other hallmarks. Dysfunctional mitochondria generate elevated reactive oxygen species that contribute to genomic instability, produce mtDNA mutations that further impair ETC function in a self-amplifying cycle, and release pro-inflammatory mitochondrial damage-associated molecular patterns (mtDAMPs) — including mitochondrial DNA fragments — that activate the cGAS-STING innate immune pathway and contribute to the chronic inflammation hallmark. Addressing mitochondrial dysfunction therefore simultaneously reduces the genomic instability and chronic inflammation inputs that compound the ageing process.
The two direct nutritional interventions for the mitochondrial dysfunction hallmark are NAD+ restoration via nicotinamide riboside (NR) — addressing the substrate limitation at Complex I and the SIRT1/SIRT3 sirtuin activity that governs mitochondrial biogenesis and antioxidant enzyme activation — and CoQ10 supplementation at the therapeutic 150mg dose documented by Fotino 2013 in the American Journal of Clinical Nutrition. The Mortensen 2014 Q-SYMBIO trial in JACC Heart Failure — enrolling 420 patients with chronic heart failure and randomising them to CoQ10 300mg daily or placebo — demonstrated a 43 percent reduction in major adverse cardiovascular events at two years, with the most significant contributor being reduced cardiovascular mortality in the CoQ10 group. For the cardiac tissue that requires the highest per-gram energy production of any organ, this effect size represents direct validation of CoQ10's role in mitigating the mitochondrial dysfunction hallmark's cardiovascular consequences. The Brenner 2018 Nature Communications NR trial's confirmation of 40 to 90 percent blood NAD+ elevation provides the parallel evidence for the upstream substrate layer.
SIRT3 and Mitochondrial Antioxidant Defence
The mechanistic connection between NAD+ restoration and mitochondrial structural preservation runs through SIRT3 — the predominant mitochondrial sirtuin that NAD+ availability regulates. As established in the memory support supplements article in this series, SIRT3 deacetylates and activates MnSOD (manganese superoxide dismutase) — the enzyme responsible for dismutating the superoxide radical generated by electron leakage at Complex I and Complex III. With adequate NAD+ and thus adequate SIRT3 activity, mitochondrial superoxide is efficiently converted to hydrogen peroxide (and then to water by catalase), preventing the lipid peroxidation chain reactions that damage the inner mitochondrial membrane and the enzyme complexes themselves. NAD+ depletion reduces SIRT3 activity, impairs MnSOD activation, and allows mitochondrial superoxide accumulation — creating the progressive self-reinforcing mitochondrial DNA damage cycle that the hallmarks framework identifies as a primary driver of accelerated cellular ageing. Reversa NR (AUST L 520794) addresses this through the NR-NAD+-SIRT3-MnSOD axis, while UbiQ Forte (AUST L 520795) provides the CoQ10 ubiquinol electron carrier that reduces the rate of superoxide generation by preventing electron leakage from semi-reduced ubisemiquinone at Complex III. The combination simultaneously addresses both the antioxidant defence layer (SIRT3-MnSOD) and the source-reduction layer (CoQ10 carrier efficiency) in mitochondrial superoxide management.
Cellular Senescence, SASP, and the Inflammation of Ageing
Cellular senescence is the hallmark that most directly explains the progressive, multi-organ systemic decline that characterises biological ageing in the absence of any single catastrophic disease event. Senescent cells — cells that have entered a stable cell cycle arrest in response to DNA damage, telomere shortening, oxidative stress, or oncogenic signalling — accumulate progressively in tissues with age because the immune surveillance mechanisms (NK cell cytotoxicity, macrophage-mediated clearance) that normally remove them become less efficient as the immune system itself ages. The accumulation of senescent cells in tissues produces the senescence-associated secretory phenotype (SASP): a continuous secretion of pro-inflammatory cytokines (TNF-α, IL-1β, IL-6), chemokines (CXCL1, CXCL2, MCP-1), growth factors (VEGF, HGF), and matrix metalloproteinases (MMP-1, MMP-3, MMP-9) that collectively create the chronic, low-grade inflammatory environment — "inflammageing" — that underpins the accelerated risk for cardiovascular disease, metabolic dysfunction, cognitive decline, and cancer that characterises aged tissue.
NF-κB is the master transcription factor governing most SASP gene expression. The p65/p50 NF-κB dimer, when released from its IκB inhibitor by IKK-β kinase phosphorylation (which SASP cytokines and DNA damage signalling both activate), translocates to the nucleus and simultaneously drives the transcription of the cytokines, MMPs, and chemokines that constitute the SASP. Curcumin's direct IKK-β inhibition — reducing the phosphorylation of IκB that would otherwise release the NF-κB dimer — provides a transcription-level brake on SASP gene expression in senescent cells that is both mechanistically specific and clinically accessible through the BioPerine-enhanced tissue delivery of CurcuNova (AUST L 520796). The 2,000 percent bioavailability enhancement documented by Shoba 1998 in Planta Medica is not merely a pharmacokinetic detail — it is the prerequisite for achieving the intracellular curcumin concentrations at which IKK-β inhibition produces the SASP suppression effect demonstrated in cell-based research. Without BioPerine, tissue-level curcumin is negligible and the NF-κB/SASP mechanism is not engaged.
Resveratrol, SIRT1, and Senescent Cell Clearance
Complementary to curcumin's SASP suppression is resveratrol's SIRT1-mediated approach to the senescence burden itself — not suppressing the SASP signal from existing senescent cells but facilitating the clearance of the senescent cells through SIRT1's role in apoptosis regulation and autophagy activation. SIRT1 deacetylates p53 — the tumour suppressor protein that governs the apoptotic fate decision — at lysine 382, which modulates p53's transcriptional activity in a context-dependent manner that can promote apoptosis in senescent cells where unresolvable DNA damage has accumulated. Additionally, SIRT1's activation of autophagy through the FOXO3a pathway provides the cellular quality control mechanism that removes damaged organelles (including dysfunctional mitochondria in the process of mitophagy) that would otherwise perpetuate the SASP-generating cellular stress state. The SIRT1-SASP connection makes the resveratrol components of both CurcuNova (150mg resveratrol) and Reversa NR (75mg resveratrol alongside NR for the NAD+ substrate) synergistically relevant to the senescence hallmark alongside the NF-κB-SASP mechanism of curcumin — two complementary approaches to the same hallmark through convergent but distinct mechanisms.
Epigenetic Drift and Nutrient Sensing Dysregulation
Two of the hallmarks of ageing that receive the least attention in conventional longevity supplement discussions but that represent the most fundamental regulatory disruptions of cellular biology with age are epigenetic alterations (the progressive drift from the epigenetic programmes established in development and early adulthood) and deregulated nutrient sensing (the dysregulation of the mTOR, AMPK, insulin/IGF-1, and sirtuin signalling pathways that govern how cells respond to nutrient availability). Both of these hallmarks are directly relevant to the SIRT1-activating compounds in the Zenutri Longevity Plus Bundle — and both are substantially more consequential for long-term healthspan than the more immediately measurable outcomes like energy levels and inflammatory markers that dominate supplement marketing.
Epigenetic Drift: The Methylation Clock and SIRT1 Histone Maintenance
The epigenetic alterations hallmark encompasses two distinct phenomena. The first is DNA methylation drift — the progressive divergence of DNA methylation patterns from their youthful state, measurable as the "epigenetic clock" developed by Horvath (confirmed to predict biological age more accurately than chronological age) and associated with both increased cancer risk (hypomethylation of previously silenced oncogenes) and reduced gene expression fidelity (hypermethylation of tumour suppressor gene promoters). The second is histone modification drift — the progressive loss of the histone acetylation, methylation, and ubiquitination marks that regulate chromatin accessibility and transcription factor access to specific gene promoters, producing a genome-wide shift toward transcriptional dysregulation and the activation of repetitive element transcription that contributes to genomic instability. SIRT1's role in epigenetic maintenance operates primarily through its deacetylase activity on histones H3K9ac, H3K14ac, and H4K16ac — marks whose removal (via SIRT1-mediated deacetylation) recruits additional repressive chromatin factors and maintains the heterochromatin domains that silence transposable elements and preserve the transcriptional programmes of differentiated cell identity. When NAD+ declines, and SIRT1 activity falls, these heterochromatin domains become progressively derepressed — a specific epigenetic consequence of NAD+ depletion that makes the NAD+-SIRT1 axis an epigenetic maintenance mechanism as well as a metabolic and mitochondrial one. The resveratrol-SIRT1 activation in both CurcuNova (AUST L 520796) and Reversa NR (AUST L 520794), combined with the NAD+ substrate restoration from NR in Reversa NR, addresses the epigenetic drift hallmark through two convergent pathways: enhanced SIRT1 catalytic activity from resveratrol's allosteric activation, and restored NAD+ availability for SIRT1's NAD+-consuming deacetylase reactions.
mTOR, AMPK, and the Nutrient Sensing Dysregulation Hallmark
The deregulated nutrient sensing hallmark reflects a systematic shift — in the Western dietary pattern and with ageing — toward chronically elevated anabolic signalling (insulin/IGF-1, mTOR) and underactivated catabolic resilience signalling (AMPK, sirtuins). mTOR (mechanistic target of rapamycin), the master anabolic nutrient sensor, promotes protein synthesis and cell growth when amino acids and growth factors are abundant. Its chronic overactivation — in the context of caloric excess and constant nutrient availability — suppresses the autophagy and protein quality control processes that remove the damaged, misfolded, and aggregated proteins that accumulate in ageing cells and are the substrate of the loss of proteostasis hallmark. Rapamycin (a direct mTOR inhibitor) consistently extends lifespan in every model organism tested — including the most clinically relevant mammalian models — specifically because mTOR inhibition restores the autophagy that nutrient excess has chronically suppressed. For nutritional supplements, the most clinically relevant intervention for the mTOR/autophagy axis is not direct mTOR inhibition (not achievable safely through oral supplementation) but the activation of AMPK — the energy deficit sensor that inhibits mTOR through TSC2 phosphorylation and directly activates autophagy through ULK1 phosphorylation. Resveratrol's SIRT1 activation produces indirect AMPK activation through the SIRT1-LKB1-AMPK pathway, providing the autophagy-restoring nutrient sensing modulation that addresses this hallmark through a mechanism that mimics aspects of the intermittent fasting and caloric restriction longevity interventions without requiring the physiological stress of those protocols. This is the mechanistic basis for resveratrol's description as a "caloric restriction mimetic" — a description that traces to the SIRT1-AMPK-autophagy pathway rather than to simplistic claims about activating longevity genes.
The Zenutri Longevity Plus Bundle: Four Pathways, Twelve Hallmarks
The Zenutri Longevity Plus Bundle ($93 AUD, save 38%) integrates four TGA AUST L-listed formulations that collectively address seven of the twelve Lopez-Otin hallmarks of ageing through distinct and non-overlapping molecular mechanisms — the most comprehensive hallmark coverage achievable within a single TGA-listed Australian-made daily supplement protocol. The architecture of the Bundle's hallmark coverage maps as follows:
Reversa NR (AUST L 520794) — NR 150mg, resveratrol 75mg, magnesium amino acid chelate 55mg, BioPerine 6.95mg.
Hallmarks addressed: Mitochondrial dysfunction (NAD+-Complex I-SIRT3-MnSOD axis); Deregulated nutrient sensing (SIRT1-LKB1-AMPK-mTOR inhibition-autophagy activation); Epigenetic alterations (NAD+-SIRT1 histone H3K9ac and H4K16ac deacetylase maintenance); Cellular senescence (SIRT1-p53 deacetylation-apoptosis modulation in senescent cells). Clinical evidence: Brenner 2018, Nature Communications — 40 to 90 percent blood NAD+ elevation at 60 days. Warfarin interaction: Reversa NR's resveratrol has mild antiplatelet activity; discuss with GP if on anticoagulant therapy.
UbiQ Forte (AUST L 520795) — CoQ10 150mg.
Hallmarks addressed: Mitochondrial dysfunction (electron carrier restoration at the Complex I/II to Complex III bottleneck, with secondary mitochondrial membrane lipid peroxidation protection in ubiquinol form); Chronic inflammation (reduced mtDAMP generation from preserved mitochondrial structural integrity, reducing cGAS-STING innate immune pathway activation). Clinical evidence: Fotino 2013, AJCN — 150mg minimum effective dose; Mortensen 2014 Q-SYMBIO, JACC Heart Failure — 43 percent MACE reduction. Anticoagulant note: GP discussion warranted before initiating if on warfarin.
CurcuNova (AUST L 520796) — Curcumin 20:1, resveratrol 150mg, activated B-complex, BioPerine 13.9mg.
Hallmarks addressed: Cellular senescence and SASP (curcumin IKK-β inhibition suppressing NF-κB-driven SASP gene expression; resveratrol SIRT1 activation facilitating senescent cell autophagy and clearance); Epigenetic alterations (resveratrol SIRT1 histone deacetylase activity; Nrf2-activated ARE antioxidant gene programme — as documented in Balogun 2003, Biochemical Journal); Chronic inflammation (curcumin NF-κB systemic anti-inflammatory action on TNF-α, IL-6, IL-1β); Loss of proteostasis (SIRT1-mediated autophagy induction supporting protein quality control). BioPerine 13.9mg provides the 2,000 per cent curcumin bioavailability enhancement from Shoba 1998, Planta Medica, which makes tissue-level curcumin engagement of these mechanisms possible. Safety: CYP3A4 piperine interaction for antidepressant users; curcumin liver warning for hepatic conditions; shellfish note for levomefolate glucosamine component.
Osteo+Core (AUST L 520792) — D3 1,000 IU (25mcg cholecalciferol), K2 MK-7 180mcg.
Hallmarks addressed: Altered intercellular communication (D3's immune cell VDR-mediated regulation of T-cell and macrophage function, modulating the systemic inflammatory signalling between immune and non-immune tissues that is a primary altered intercellular communication mechanism); and the bone and vascular biology that the hallmarks framework does not explicitly classify but that is directly relevant to Australian adult healthspan — MK-7-activated osteocalcin for bone mineral density maintenance (the Knapen 2013 three-year RCT in Osteoporosis International confirming 180mcg MK-7 improves bone mineral density), and MK-7-activated matrix Gla protein (MGP) for vascular calcification prevention (the Geleijnse 2004 cohort in the Journal of the American College of Cardiology confirming 57 percent cardiac mortality reduction in the highest K2 intake quartile). Note: Osteo+Core's K2 component carries an anticoagulant interaction note for warfarin users — discuss with GP before initiating if on anticoagulant therapy.
The Optimal Timing Window: Why the 40s Are the Most Impactful Entry Point
The hallmarks framework provides a precise answer to one of the most common questions about longevity supplementation: when to start. Each hallmark has a window in which it is most effectively addressed — the period in which it is accumulating but has not yet produced irreversible structural changes. NAD+ decline is measurably occurring from the late 30s but has not yet produced the structural neurological changes of late-life cognitive impairment. The SASP burden is increasing from the 40s but not yet sufficient to dominate tissue inflammatory biology. Epigenetic drift is accumulating across the adult lifespan but remains partially reversible through SIRT1 maintenance during the decades before cell identity loss is structural. Bone mineral density loss from D3 and K2 insufficiency is beginning in the 40s but the MK-7 protocol produces the greatest relative benefit when initiated before significant trabecular bone structure loss. The 40 to 55-year bracket is the optimal longevity supplement initiation window precisely because it is the period in which all four addressable hallmarks are accumulating at their most modifiable rates, which is why the Longevity Plus Bundle is designed as a proactive healthspan investment rather than a reactive response to established disease. Every decade of the hallmark mitigation that the Bundle provides during the modifiable window produces compounding biological protection that becomes progressively harder to achieve once structural hallmark changes are established.
Connecting the Longevity Bundle to the Complete Zenutri System
The Longevity Plus Bundle's four-formulation hallmark coverage is complemented by the other formulations in the Zenutri range that address the remaining hallmarks. C E B Optima (AUST L 521487) contributes to the genomic instability hallmark through Vitamin C's DNA repair enzyme co-factor activity and natural mixed tocopherols' lipid peroxidation protection of nuclear and mitochondrial DNA. Immunaxis (AUST L 521494) contributes to the stem cell exhaustion hallmark through zinc glycinate's role in haematopoietic stem cell proliferation and selenomethionine's GPx4 protection of stem cell niches from oxidative damage. MagLipo Core (AUST L 520793) provides the ALA-PDH and magnesium-ATP layer that underpins the mitochondrial efficiency that the Longevity Bundle's NAD+ and CoQ10 components build upon. The complete Zenutri product range, assessed through the hallmarks framework, represents the most comprehensively evidence-grounded nutritional approach to the biology of healthspan extension available in TGA-listed, Australian-made, individually AUST L-registered form. For a personalised recommendation on which combination of formulations best addresses your specific hallmark profile based on your age, health history, and symptom cluster, take the free Zenutri health quiz.
Longevity Is Not What Happens to You — It's What You Maintain
The hallmarks of ageing framework has transformed longevity from an aspirational concept into a mechanistic programme with specific molecular targets, specific interventions, and specific windows of maximum modifiability. The Australian adult in their 40s who begins a multi-pathway longevity supplement protocol addressed to mitochondrial dysfunction, cellular senescence, epigenetic drift, and nutrient sensing dysregulation is not taking supplements in the vague hope of living longer. They are providing specific molecular co-factors and signalling modulators to cellular systems that the peer-reviewed biology of ageing has identified as primary determinants of healthspan — the years of life spent functioning at full cognitive, cardiovascular, musculoskeletal, and metabolic capacity.
The Zenutri Longevity Plus Bundle represents the most cohesive and evidence-grounded four-formulation longevity protocol available in the Australian TGA-listed supplement market. Every ingredient form traces to the bioavailability hierarchy. Every dose traces to a clinical trial. Every hallmark addressed traces to a specific molecular mechanism. Every AUST L number traces to a verifiable TGA ARTG registration confirming Australian cGMP manufacture and label accuracy.
Explore the Zenutri Longevity Plus Bundle — or take the free health quiz to receive a personalised recommendation mapping your specific age, health history, and biological priorities to the right protocol.
The twelve hallmarks of ageing are accumulating right now. Four of them are nutritionally modifiable. The window in which that modification is most impactful is the decade you are currently in.
Frequently Asked Questions
What are the best longevity supplements in Australia?
The longevity supplements with the strongest combined evidence across the hallmarks of ageing framework are: nicotinamide riboside (NR) for NAD+-SIRT1-SIRT3 mitochondrial function, nutrient sensing, and epigenetic maintenance (Brenner 2018, Nature Communications); CoQ10 at 150mg for electron transport chain function and the Q-SYMBIO-confirmed cardiovascular protection (Mortensen 2014, JACC Heart Failure); curcumin with BioPerine for NF-κB-SASP inhibition from senescent cells (Shoba 1998 bioavailability); resveratrol for SIRT1 epigenetic maintenance and mTOR/AMPK nutrient sensing modulation; and D3/MK-7 K2 for bone mineral density (Knapen 2013) and vascular calcification prevention (Geleijnse 2004). These are combined in the Zenutri Longevity Plus Bundle.
What are the hallmarks of ageing and why do they matter for supplement selection?
The twelve hallmarks of ageing — genomic instability, telomere attrition, epigenetic alterations, loss of proteostasis, deregulated nutrient sensing, mitochondrial dysfunction, cellular senescence, stem cell exhaustion, altered intercellular communication, disabled macroautophagy, chronic inflammation, and dysbiosis — are the cellular and molecular mechanisms that Lopez-Otin and colleagues identified in Cell (2013) as the converging biological explanations for why organisms age. They matter for supplement selection because they identify which molecular mechanisms a given compound actually targets — distinguishing supplements that modulate a genuine hallmark mechanism from those that produce a correlational health benefit without directly addressing the ageing biology. NAD+ precursors address mitochondrial dysfunction and deregulated nutrient sensing. Curcumin and resveratrol address cellular senescence and SASP. The D3/K2 combination addresses the calcium metabolism consequences of altered intercellular communication in bone and vascular tissue. A supplement claiming "longevity support" without identifying which hallmark it addresses is making an unverifiable marketing claim rather than a mechanistic claim.
What is cellular senescence and how do supplements address it?
Cellular senescence is a stable cell cycle arrest that damaged cells enter when DNA damage signalling cannot be resolved. Senescent cells accumulate in tissues with age and secrete the senescence-associated secretory phenotype (SASP) — a pro-inflammatory mixture of TNF-α, IL-1β, IL-6, MMP-1, MMP-3, MMP-9, and VEGF that progressively impairs the tissue function of every organ system. NF-κB is the master transcription factor governing most SASP gene expression. Curcumin's IKK-β inhibition reduces IκB phosphorylation and NF-κB nuclear translocation, suppressing SASP cytokine transcription directly. Resveratrol's SIRT1 activation promotes senescent cell autophagy and apoptosis via FOXO3a and p53 deacetylation pathways, reducing senescent cell accumulation and thereby attenuating the SASP signal. Together — as co-formulated in CurcuNova (AUST L 520796) — these two mechanisms address the senescence hallmark from both the SASP suppression and the senescent cell clearance dimensions.
How does the mTOR pathway relate to longevity?
mTOR (mechanistic target of rapamycin) is the master anabolic nutrient-sensing kinase that promotes protein synthesis and inhibits autophagy when nutrients are abundant. Chronic mTOR overactivation — the state produced by the caloric excess of the Western dietary pattern — suppresses the autophagy and protein quality control that remove the damaged proteins and organelles accumulating in ageing cells, driving the loss of proteostasis hallmark. Rapamycin (a direct mTOR inhibitor) extends lifespan in every tested model organism, specifically by restoring autophagy. For oral supplementation, the most accessible mTOR-modulating pathway is the SIRT1-LKB1-AMPK cascade: resveratrol activates SIRT1, which activates LKB1 kinase, which phosphorylates and activates AMPK, which inhibits mTOR through TSC2 phosphorylation and directly activates autophagy through ULK1 phosphorylation. This indirect mTOR inhibition — via the SIRT1-AMPK axis — is the mechanistic basis for resveratrol's caloric restriction mimetic designation and its relevance to the deregulated nutrient sensing and loss of proteostasis hallmarks simultaneously.
Is it too early to start taking longevity supplements in your 40s?
The 40s represent the optimal time to begin a longevity supplement protocol — not too early, and critically, not after the modifiable window has narrowed. NAD+ decline is measurably occurring from the late 30s, but structural consequences are minimal at 42. Senescent cell burden is increasing but autophagic and immune clearance mechanisms still respond meaningfully to SIRT1 and NF-κB modulation. Epigenetic drift is accumulating but remains substantially reversible through SIRT1 histone maintenance. Bone mineralisation loss is beginning, but the MK-7 RCT evidence demonstrates the greatest relative benefit from initiating the D3/K2 protocol before significant trabecular bone loss is established. The hallmarks framework's insight — that each hallmark has a window of maximum modifiability before structural changes make intervention less effective — positions the 40s as the highest-leverage entry point for longevity-oriented nutritional supplementation, making the Zenutri Longevity Plus Bundle a proactive healthspan investment rather than a reactive management tool for established decline.
Ready to take action on your health?






Leave a comment
This site is protected by hCaptcha and the hCaptcha Privacy Policy and Terms of Service apply.